Ayzeze: Tento produkt je zdravotnický prostředek, který musí být používán pouze k určenému účelu a v souladu s pokyny k použití.
Tento prostředek nenahrazuje odborné lékařské poradenství, diagnózu ani léčbu. V případě pochybností se před použitím poraďte s kvalifikovaným zdravotnickým odborníkem. Prodejce/distributor nenese odpovědnost za škody způsobené nesprávným použitím, neoprávněnými úpravami nebo použitím jiným než podle určeného účelu.
-----
Please be sure to read the instruction manual.
Generic Name
Pulse Oximeter
Product Name
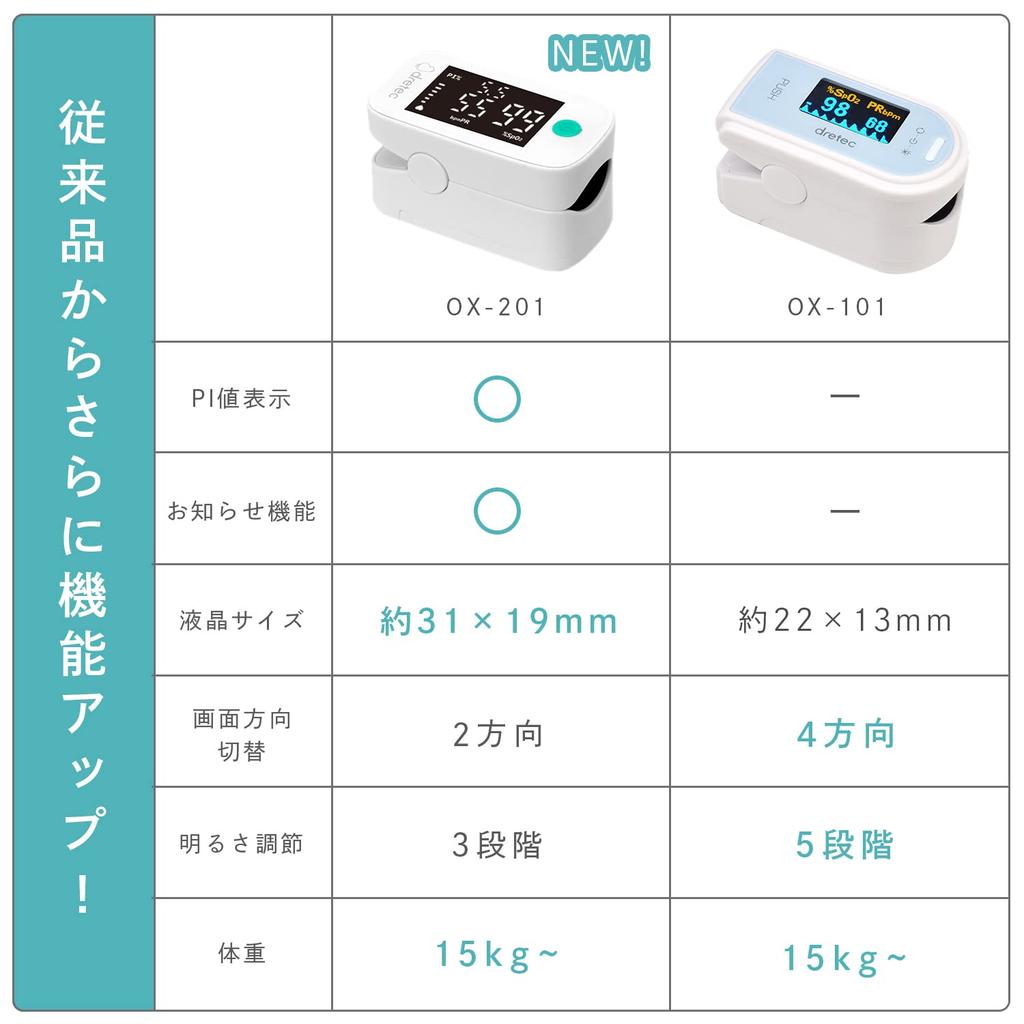
Pulse Oximeter OX-201
Warning
- Do not use inside or in conjunction with a hyperbaric oxygen device. (This may cause malfunction, damage, or failure.)
- Do not use in the presence of flammable substances (anesthetic gas, high-concentration oxygen, gasoline, etc.) or flammable vapors. (This may cause a fire.)
Contraindications/Prohibitions
Intended Use (Patients)
- This device is intended for patients weighing 15 kg or more and with a finger thickness of 8 mm or more. Do not use on infants or young children under these ages.
- If you use a pacemaker, consult your doctor before using a pulse oximeter.
Concomitant Medical Devices
- Do not use in areas controlled by radiation equipment or MRI (magnetic resonance imaging) equipment. (This may cause malfunction, damage, or failure.)
How to Use
- Do not wear the device for long periods of time or continuously.
- Do not wear on injured fingers.
Product Specifications
[Measurement Range]
SpO2: 70% - 99%
Pulse: 30 bpm - 250 bpm
PI: 0.3% - 20.0%
[Measurement Accuracy]
SpO2: 70% - 99%: ±2% (no specification for values below 70%)
Pulse: 30 bpm - 100 bpm: ±2 bpm, 100 bpm - 250 bpm: ±2%
PI: 0.3% - 1.0%: ±0.2%, 1.1% - 20.0%: ±2.0%
Precautions for Use
(1) Conditions of Use and Basic Precautions
① Read the instruction manual before use.
② Do not use for purposes other than measuring blood oxygen saturation, pulse rate, and perfusion index.
③ Do not repair or modify.
④ Do not use on anything other than your fingers.
⑤ Keep the finger clean.
(2) Precautions During Use
① If a malfunction is detected in the device, discontinue use immediately.
② If you notice any abnormalities in your body or the device, discontinue use immediately.
③ Do not use simultaneously with medical devices such as electrosurgical devices.
④ Do not move your finger or body during measurement.
⑤ Measurement may not be accurate in the following cases:
・When there is a high amount of abnormal hemoglobin (such as carboxyhemoglobin)
・When pigments (such as methylene blue) are present in the blood
・When blood circulation at the measurement site is insufficient
・When the device is worn on the same arm as the blood pressure monitor
・When ambient light, such as lighting, fluorescent light, or direct sunlight, is too strong
・When you have nail polish or hand cream on your nails
・When the device is not worn correctly on your finger
・When you are moving vigorously
⑥ Do not pull on the device by the strap.
(3) Precautions After Use
① After use, wipe the finger holder with a cloth soaked in rubbing alcohol.
② To clean, wipe with a soft cloth dampened with water or mild detergent, then dry with a dry cloth.
Maintenance and Inspection Information
(1) If you notice any malfunctions or abnormalities with this product, contact the appropriate contact information.
(2) When using this product after a period of inactivity, verify that it is operating normally and safely before use.
Name of Manufacturer and Distributor
Manufacturer: Doretec Co., Ltd.
Manufacturer: Shenzhen Yimi Life Technology Co., Ltd.
Country: People's Republic of China
Contact Information
Doretec Co., Ltd. Customer Service Center
2-3-9 Ryutsu Danchi, Koshigaya City, Saitama Prefecture 343-0824
Tel: 0120-875-019 (Mon-Fri 9:00-12:00, 13:00-17:00, excluding public holidays and company-designated holidays)
Purpose of Use or Effects/Efficacy
Measure and display transcutaneous arterial blood oxygen saturation.
Medical Device Classification: Special Maintenance Management Medical Device/Special Management Medical Device
Medical Device Certification Number: 304AKBZX00071000